Video Lesson
1. What Is a Mole?
Chemistry calculations often involve numbers that are unimaginably large. Atoms, molecules and ions are so small that even a tiny sample of a substance contains an enormous number of particles. The idea of the mole gives us a practical way to count these particles without having to list them one by one.
A mole is a unit of amount in chemistry. When we say “1 mole”, we mean a fixed number of particles. That number is always the same, regardless of the substance being discussed.
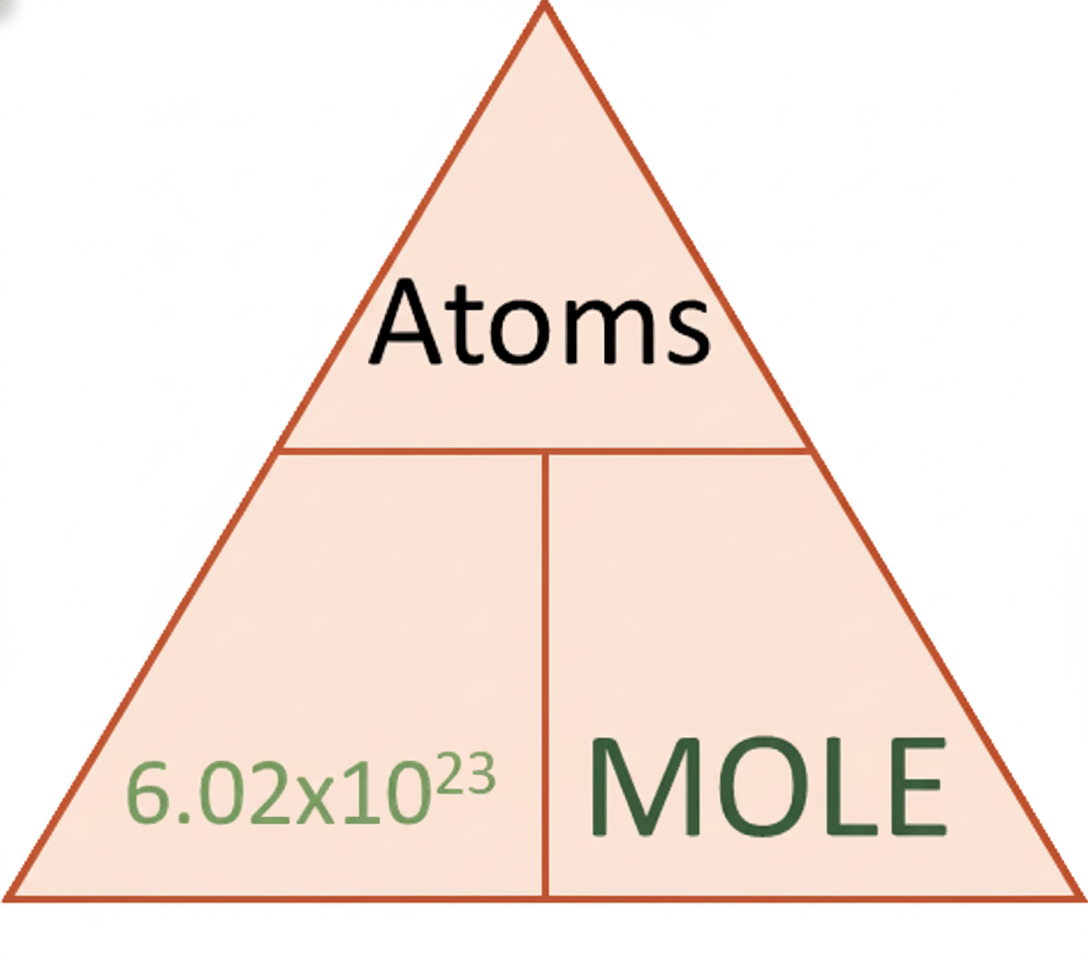
1 mole = 6.02 × 1023 particles
This number is called Avogadro’s constant. For example, 1 mole of magnesium contains 6.02 × 1023 magnesium atoms, and 1 mole of water also contains 6.02 × 1023 water molecules.
Important Mole Facts
- One mole of any substance contains 6.02 × 1023 particles (atoms, molecules or ions).
- One mole of any element has a mass equal to its relative atomic mass in grams.
- 12 g of carbon therefore contains 1 mole of carbon atoms.

2. Linking Moles to Particles
Once you know that 1 mole contains 6.02 × 1023 particles, you can convert between moles and number of particles using a direct formula:
number of particles = moles × 6.02 × 1023
In words, this means that the number of particles increases in direct proportion to the number of moles. Half a mole contains half as many particles, and four moles contain four times as many particles.
Worked Examples
Q. How many atoms are in half a mole of magnesium?
A. 0.5 × 6.02 × 1023 = 3.01 × 1023 atoms
Q. How many moles are represented by 6.02 × 1022 sodium atoms?
A. 6.02 × 1022 ÷ 6.02 × 1023 = 0.1 mol
Q. How many atoms are there in 4 moles of iron?
A. 4 × 6.02 × 1023 = 2.408 × 1024 atoms
Q. Which has more atoms: 1 mole of copper or 1 mole of lead?
A. Neither. Both contain the same number of atoms because 1 mole of any element contains 6.02 × 1023 atoms.
Q. How many water molecules are in 0.4 moles of H2O?
A. 0.4 × 6.02 × 1023 = 2.408 × 1023 molecules
3. Linking Moles to Mass
For elements, 1 mole has a mass equal to the relative atomic mass in grams. This is why the periodic table becomes useful in mole calculations.
For example, carbon has a relative atomic mass of 12, so 1 mole of carbon has a mass of 12 g. Helium has a relative atomic mass of 4, so 2 moles of helium have a mass of 8 g.
For an element: 1 mole = relative atomic mass in grams
This relationship tells you that heavier atoms will have a greater mass per mole. One mole of lead therefore weighs more than one mole of gold, because lead has the larger relative atomic mass (207 compared with 197).
Worked Examples
Q. If 12 g of carbon represents 1 mole of carbon, how much does half a mole of carbon weigh?
A. 0.5 × 12 = 6 g
Q. What does a tenth of a mole of calcium weigh?
A. 0.1 × 40 = 4 g
Q. What weighs more, 1 mole of lead or 1 mole of gold?
A. 1 mole of lead, because lead has the larger relative atomic mass (207 compared with 197).
Q. What does 2 moles of helium weigh?
A. 2 × 4 = 8 g
Q. How many moles are found in 21 g of krypton?
A. 21 ÷ 84 = 0.25 mol
4. Deriving the Mole Equation
After linking moles to mass, we can derive a general equation. If the mass of 1 mole equals the relative atomic mass in grams, then the mass for any number of moles can be found by multiplying the relative atomic mass by the number of moles:
mass = RAM × moles
Worked Example
Helium has a relative atomic mass of 4. For 2 moles of helium: mass = 4 × 2 = 8 g.
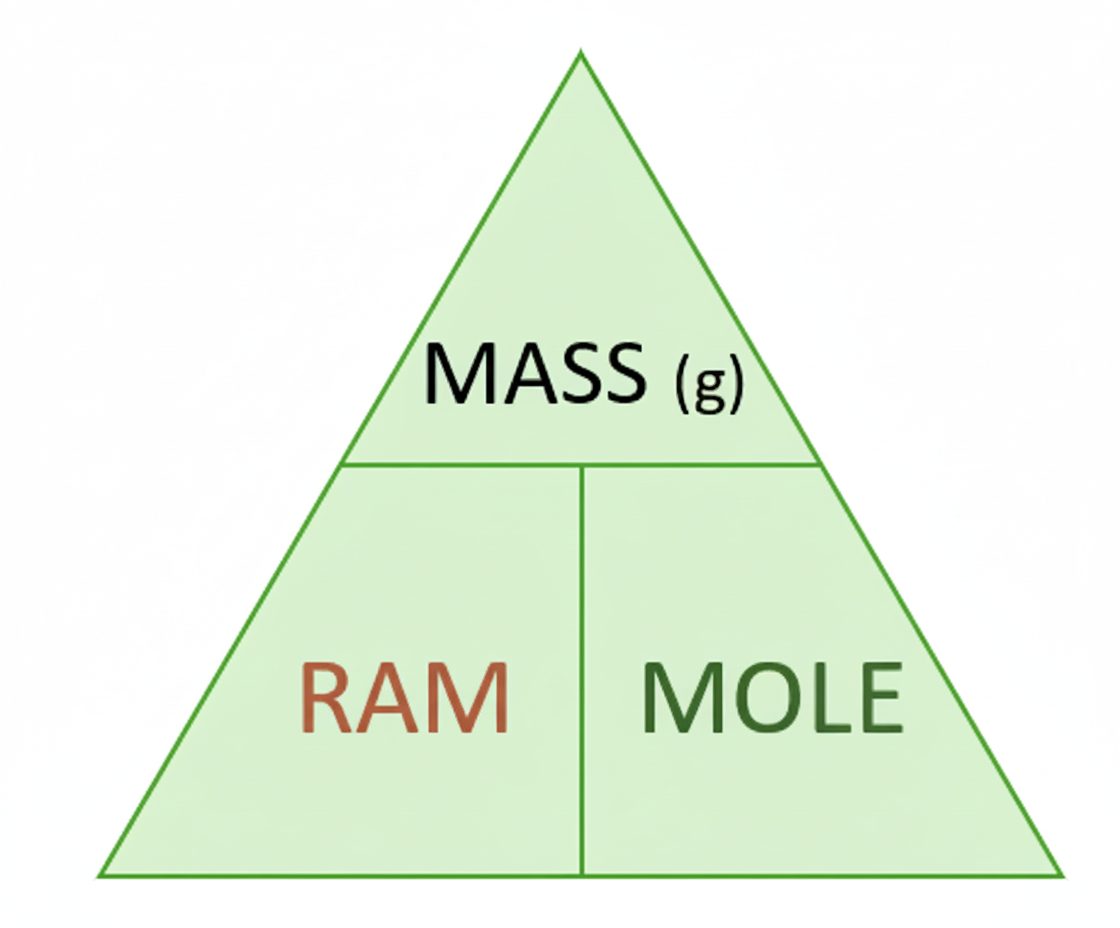
The triangle is a useful memory aid. Put mass at the top, and RAM and moles underneath. If you need mass, multiply the two bottom values. If you need moles, divide mass by RAM.
5. Extending the Idea to Compounds
The same rule can be used for compounds and molecules. The only difference is that, for a compound, you use relative formula mass instead of relative atomic mass.
Relative formula mass is found by adding together the relative atomic masses of every atom shown in the formula. This means subscripts matter, because they tell you how many of each atom are present.
Relative formula mass = all the RAM values added according to the chemical formula
Example
Na2CO3: 23 + 23 + 12 + (16 × 3) = 106
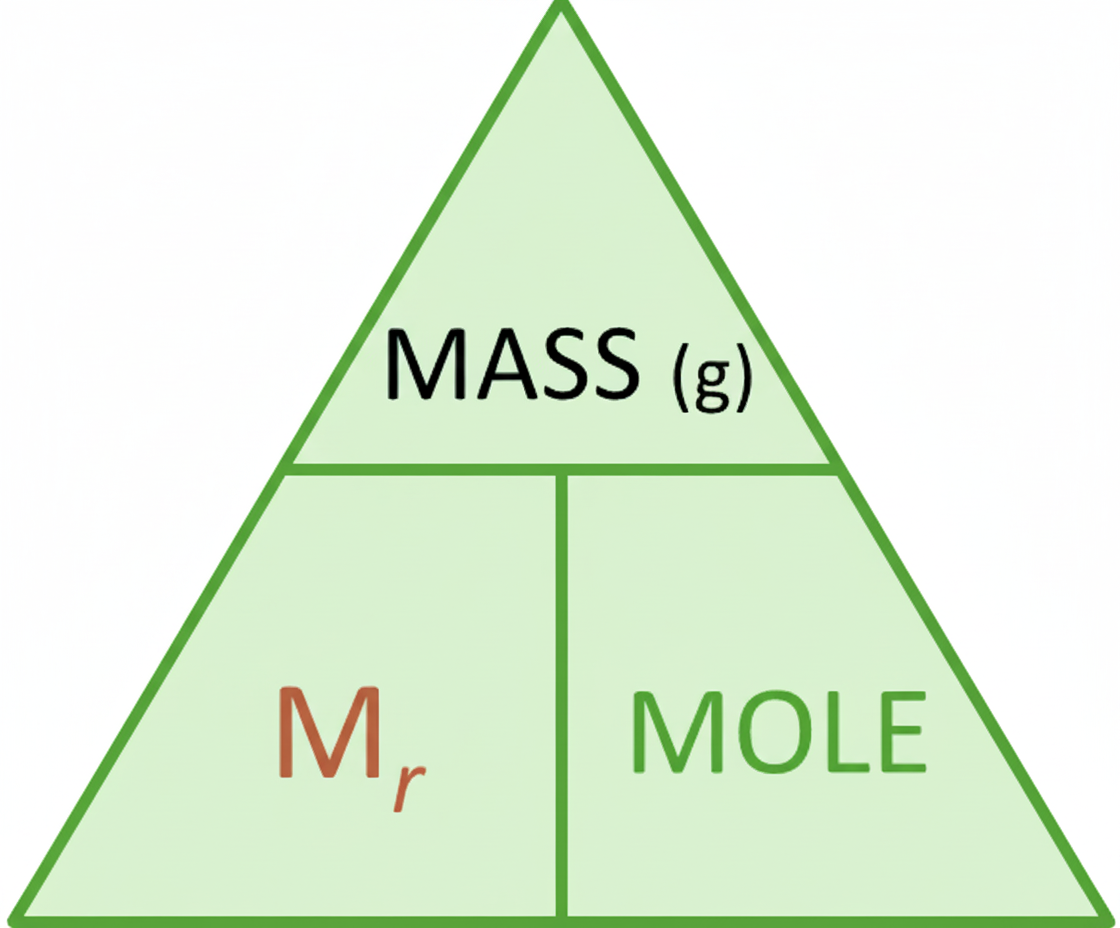
This means the relative formula mass of sodium carbonate is 106. Once that value has been found, you can use it in the same way that you used relative atomic mass for an element. To make the notation easier, we replace RAM or RFM with one symbol: Mr.
Using Mr means one equation works for both elements and compounds:
mass = Mr × moles
moles = mass ÷ Mr
The memory phrase is that “Mr Mole wears a MASSive hat!”, reminding you that MASS sits above Mr and MOLES in the formula triangle.

6. Using the Mole Equation in Practice
Let’s bring everything together. Some questions ask you to find moles from mass, some ask you to find mass from moles, and some combine mole calculations with Avogadro’s constant to work out the number of particles.
A good method is to ask yourself three questions each time: what am I given, what am I trying to find, and which equation connects those two quantities? This helps prevent using the wrong formula.
Worked Examples
Q. How many moles are in 25 g of silver?
A. 25 ÷ 108 = 0.231... mol, so approximately 0.23 mol
Q. How many atoms are in a 10 g sample of calcium?
A. First find moles: 10 ÷ 40 = 0.25 mol. Then number of atoms = 0.25 × 6.02 × 1023 = 1.505 × 1023 atoms
Q. How much does 0.25 moles of NaCl weigh in grams?
A. Mr of NaCl = 23 + 35.5 = 58.5, so mass = 58.5 × 0.25 = 14.625 g
Q. How many moles are in 2.5 g of sugar, C6H12O6?
A. Mr = (12 × 6) + (1 × 12) + (16 × 6) = 180, so moles = 2.5 ÷ 180 = 0.0139... mol, about 0.014 mol
Q. How many molecules of water are in a 150 g sample of H2O?
A. First find moles: 150 ÷ 18 = 8.33... mol. Then molecules = 8.33 × 6.02 × 1023 = 5.02 × 1024 molecules
7. Summary
- A mole is a counting unit used for extremely large numbers of particles.
- 1 mole always contains 6.02 × 1023 particles.
- For elements, 1 mole has a mass equal to the relative atomic mass in grams.
- For compounds, use Mr (relative formula mass) instead of relative atomic mass.
- To find particles from moles, multiply by 6.02 × 1023.
- To find moles from mass, divide by Mr.
- Always show units clearly and decide first whether the question is about mass, moles or particles.
Related Lessons
- Balancing Symbol Equations – practise counting atoms in formulae, which you will need for mole calculations.
- Ions & Chemical Formulae – learn how to write ionic compound formulae using the charges on ions.