Video Lesson
Introduction
An important part of GCSE Chemistry is using ions to work out chemical formulae. Before we learn how to do this, we need to understand the different types of ions. There are three groups to get to grips with:
- The ions of elements that can be worked out from the element’s group number
- The ions of transition metal elements
- The molecular ions and hydrogen (these have to be learnt)
Let’s look at each group in turn.
1. Ions from Group Numbers
The periodic table can tell us the charge on many ions. Metals lose electrons to form positive ions, and non-metals gain electrons to form negative ions. The number of electrons lost or gained depends on the element’s group number:
| Group | Ion Charge | Examples |
|---|---|---|
| Group 1 | +1 | Li+, Na+, K+ |
| Group 2 | +2 | Be2+, Mg2+, Ca2+ |
| Group 3 | +3 | Al3+, Ga3+ |
| Group 5 | −3 | N3−, P3− |
| Group 6 | −2 | O2−, S2− |
| Group 7 | −1 | F−, Cl−, Br− |
For metals in Groups 1, 2 and 3, the ion charge equals the group number. For non-metals in Groups 5, 6 and 7, subtract the group number from 8 to find how many electrons are gained (giving a negative charge). You do not need to memorise individual ion charges for these elements; just use their position in the periodic table.
2. Transition Metal Ions
The transition metals form positive ions just like the other metals, but the periodic table will not tell us the charge on these ions. Instead, we find the charge from roman numerals in the compound’s name:
| Name | Ion |
|---|---|
| Iron (II) | Fe2+ |
| Iron (III) | Fe3+ |
| Chromium (III) | Cr3+ |
| Copper (II) | Cu2+ |
| Silver (I) | Ag+ |
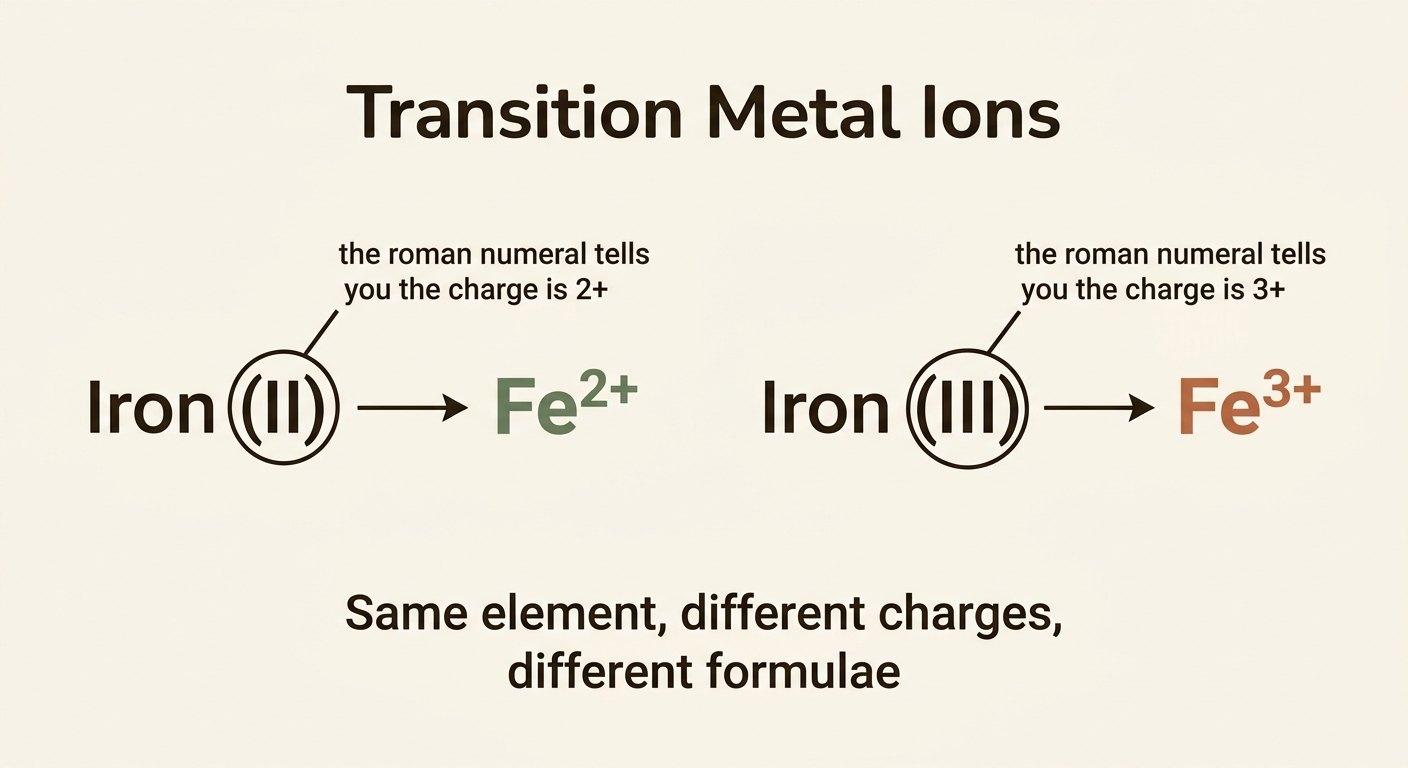
Notice that the same transition metal can form ions with different charges. Iron, for example, can be Fe2+ or Fe3+. The roman numeral in the name tells you which one is being used. If you see “iron (II)”, the iron ion has a 2+ charge. If you see “iron (III)”, it has a 3+ charge.
Always look for the roman numeral in the compound name. Without it, you cannot determine the charge on a transition metal ion.
3. Molecular Ions and Hydrogen
There are some ions that are made from more than one element. Most of these are negative. These are called molecular ions and they just have to be learnt:
| Name | Formula | Charge |
|---|---|---|
| Phosphate | PO43− | 3− |
| Sulfate | SO42− | 2− |
| Carbonate | CO32− | 2− |
| Hydroxide | OH− | 1− |
| Nitrate | NO3− | 1− |
| Hydrogen | H+ | 1+ |
| Ammonium | NH4+ | 1+ |
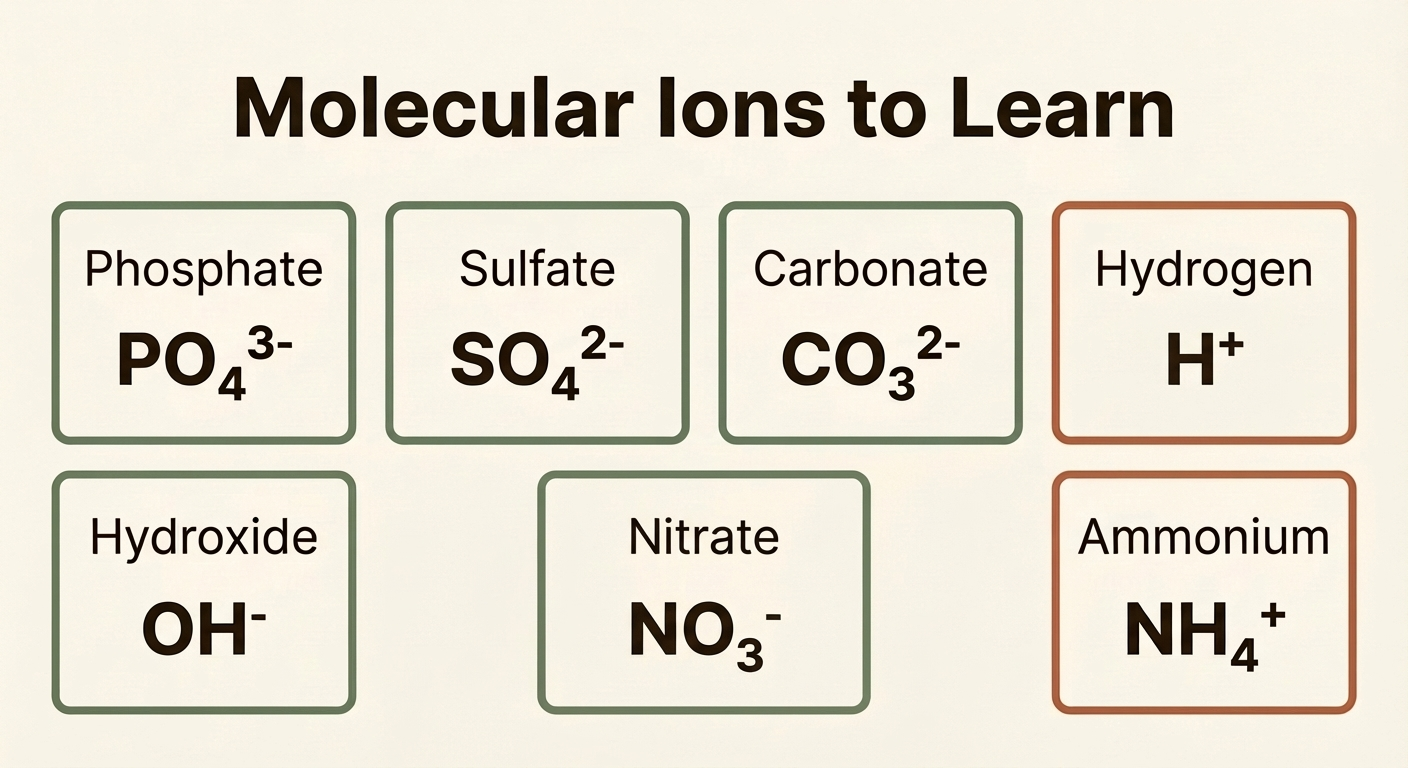
You will need to know these ions and their charges. A helpful way to remember them is to practise writing them out until they become second nature.
4. Writing Chemical Formulae from Ions
Ionic compounds are made up of positive ions and negative ions. The key rule is that the charges must cancel out to ensure the ionic compound is electrically neutral. This means the total positive charge must equal the total negative charge.
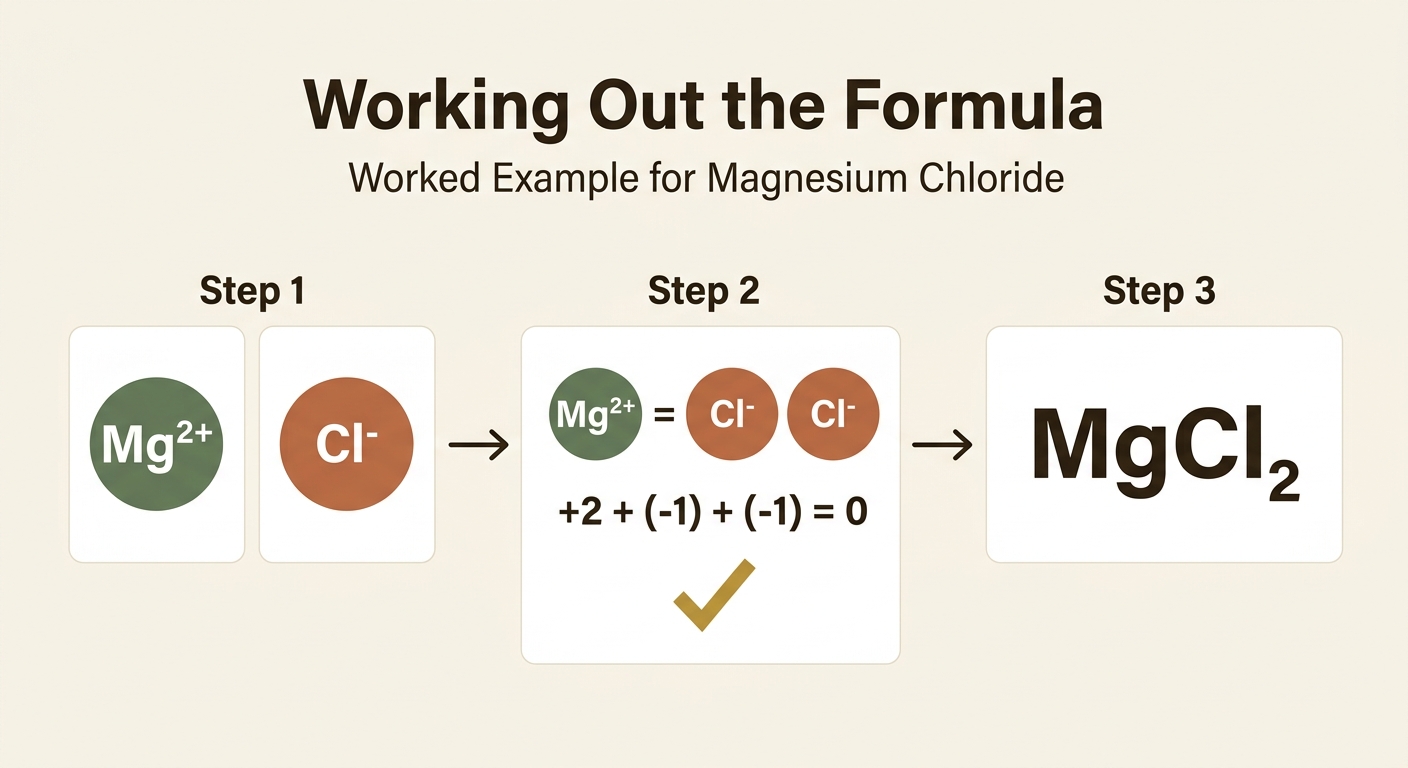
Worked Example 1: Magnesium Chloride
Step 1: Write down the ions. Magnesium is in Group 2, so it forms Mg2+. Chlorine is in Group 7, so it forms Cl−.
Step 2: Balance the charges. Magnesium has a 2+ charge, and each chloride ion has a 1− charge. We need two chloride ions to cancel the 2+ charge: (+2) + (2 × −1) = 0.
Answer: MgCl2
Worked Example 2: Aluminium Oxide
Step 1: Write down the ions. Aluminium is in Group 3, so it forms Al3+. Oxygen is in Group 6, so it forms O2−.
Step 2: Balance the charges. We need the total positive and negative charges to cancel. The smallest numbers that work are 2 aluminium ions and 3 oxide ions: (2 × +3) + (3 × −2) = +6 − 6 = 0.
Answer: Al2O3
Worked Example 3: Sodium Sulfate
Step 1: Write down the ions. Sodium is in Group 1, so it forms Na+. Sulfate is a molecular ion: SO42−.
Step 2: Balance the charges. Each sodium ion has a 1+ charge, and sulfate has a 2− charge. We need two sodium ions: (2 × +1) + (−2) = 0.
Answer: Na2SO4
Worked Example 4: Iron (II) Oxide vs Iron (III) Oxide
This example shows why the roman numeral matters. The same two elements give different formulae depending on the charge of the iron ion:
- Iron (II) oxide: Fe2+ and O2−. The charges are already equal and opposite, so the formula is simply FeO.
- Iron (III) oxide: Fe3+ and O2−. We need 2 iron ions and 3 oxide ions: (2 × +3) + (3 × −2) = 0. The formula is Fe2O3.
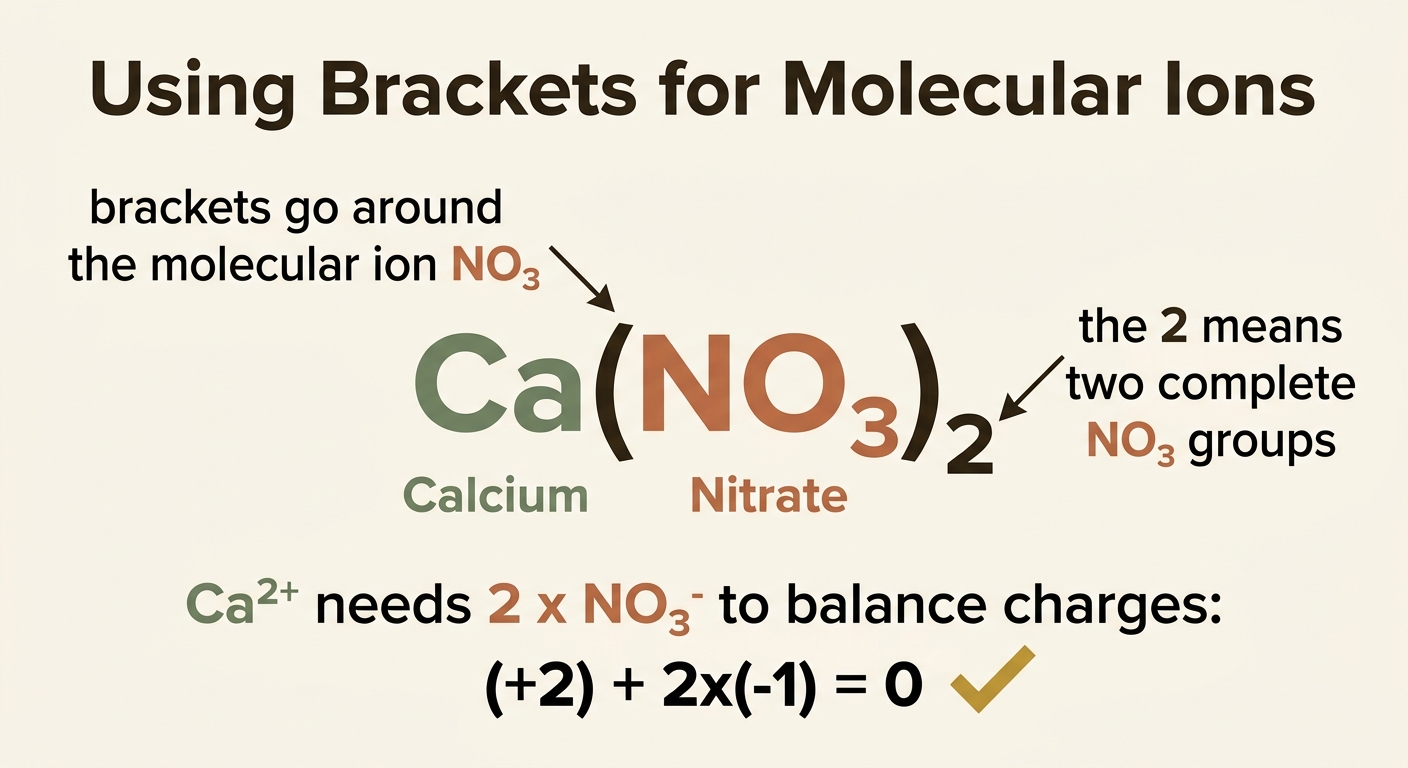
Worked Example 5: Calcium Nitrate
Step 1: Write down the ions. Calcium is in Group 2, so it forms Ca2+. Nitrate is a molecular ion: NO3−.
Step 2: Balance the charges. We need two nitrate ions to cancel the 2+ charge of calcium: (+2) + (2 × −1) = 0.
Answer: Ca(NO3)2
Notice the brackets around the nitrate ion. When you need more than one of a molecular ion, you put the ion in brackets and write the number outside. This tells us there are two complete NO3 groups, not just two oxygen atoms at the end.
Worked Example 6: Aluminium Sulfate
Step 1: Write down the ions. Aluminium forms Al3+. Sulfate is SO42−.
Step 2: Balance the charges. We need 2 aluminium ions and 3 sulfate ions: (2 × +3) + (3 × −2) = +6 − 6 = 0.
Answer: Al2(SO4)3
To work out the formula of an ionic compound: (1) write down the ions with their charges, (2) find the simplest ratio that makes the total charge zero, and (3) write the formula using subscript numbers. Remember to use brackets around molecular ions if you need more than one of them.
5. Practice Questions
Now it’s your turn! Can you work out the chemical formulae for these compounds? Try them yourself before checking the answers below.
- Lithium carbonate
- Copper (II) oxide
- Silver nitrate
- Potassium sulfate
- Aluminium bromide
- Sodium oxide
- Magnesium hydroxide
- Hydrogen sulfate (sulfuric acid)
- Barium chloride
- Beryllium nitrate
Answers
| Compound | Positive Ion | Negative Ion | Formula |
|---|---|---|---|
| Lithium carbonate | Li+ | CO32− | Li2CO3 |
| Copper (II) oxide | Cu2+ | O2− | CuO |
| Silver nitrate | Ag+ | NO3− | AgNO3 |
| Potassium sulfate | K+ | SO42− | K2SO4 |
| Aluminium bromide | Al3+ | Br− | AlBr3 |
| Sodium oxide | Na+ | O2− | Na2O |
| Magnesium hydroxide | Mg2+ | OH− | Mg(OH)2 |
| Hydrogen sulfate (sulfuric acid) | H+ | SO42− | H2SO4 |
| Barium chloride | Ba2+ | Cl− | BaCl2 |
| Beryllium nitrate | Be2+ | NO3− | Be(NO3)2 |
Summary
- Ions from Groups 1, 2 and 3 form positive ions with a charge equal to the group number. Groups 5, 6 and 7 form negative ions.
- Transition metal ion charges are shown by roman numerals in the compound name.
- Molecular ions (sulfate, carbonate, hydroxide, nitrate, phosphate, ammonium) must be memorised.
- To write a chemical formula, the positive and negative charges must cancel to give a neutral compound.
- Use brackets around molecular ions when more than one is needed in the formula.
Related Lessons
- Atomic Structure – revise how the periodic table determines the number of protons, neutrons, and electrons.
- Balancing Symbol Equations – put your formulae skills into practice by balancing chemical equations.