Video Lesson
Introduction
In chemistry, reactions are represented by symbol equations. The reactants and products are different substances, but the total number of atoms on both sides of the equation must always be equal. This follows the Law of Conservation of Mass: matter cannot be created or destroyed in a chemical reaction. It can only be rearranged into new substances.
Imagine you have a bag of Lego bricks. You can rearrange the bricks to build new things, but you cannot create new bricks or throw any away. The total number of bricks stays the same before and after, even though you have built something different:
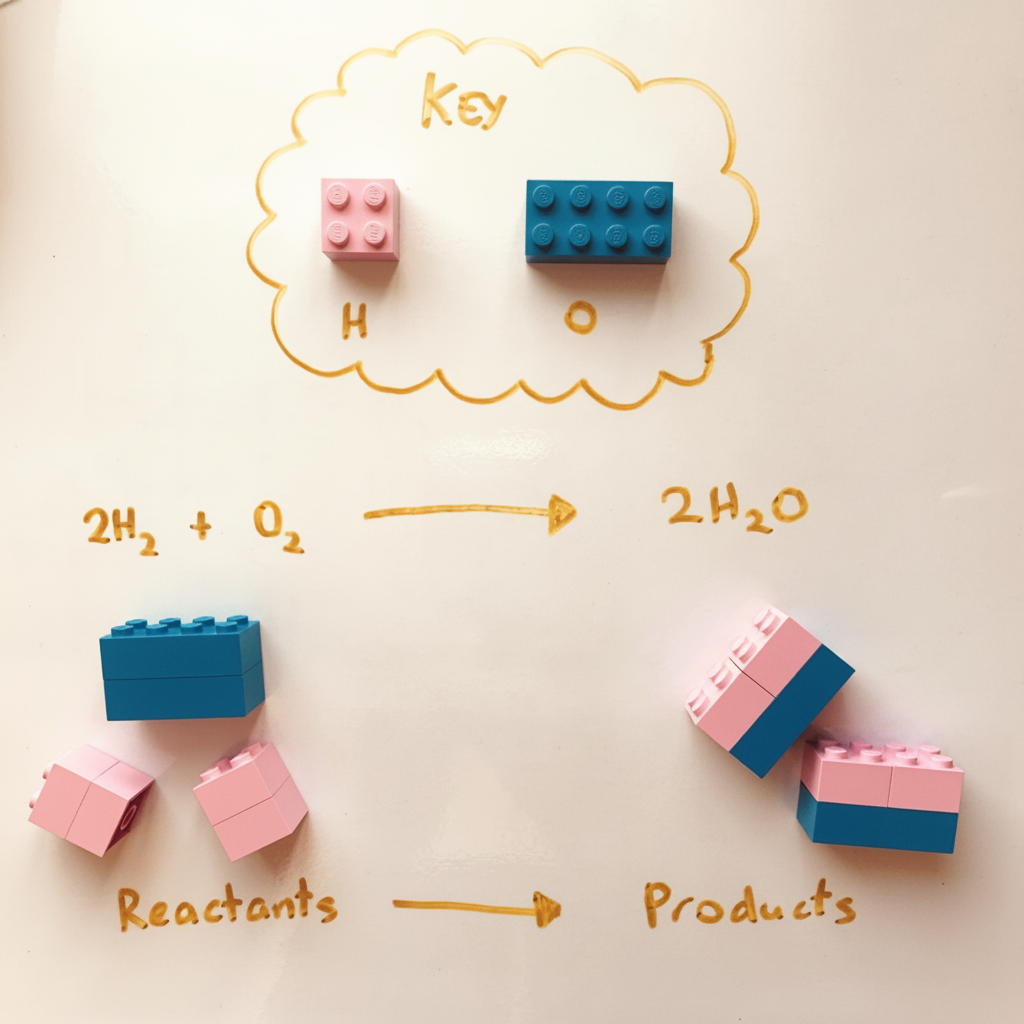
Balancing a chemical equation ensures that you always have the same number of atoms on both sides of the equation, without changing the individual chemical formulae of the substances reacting or of the new products forming. There are some important balancing rules to follow:
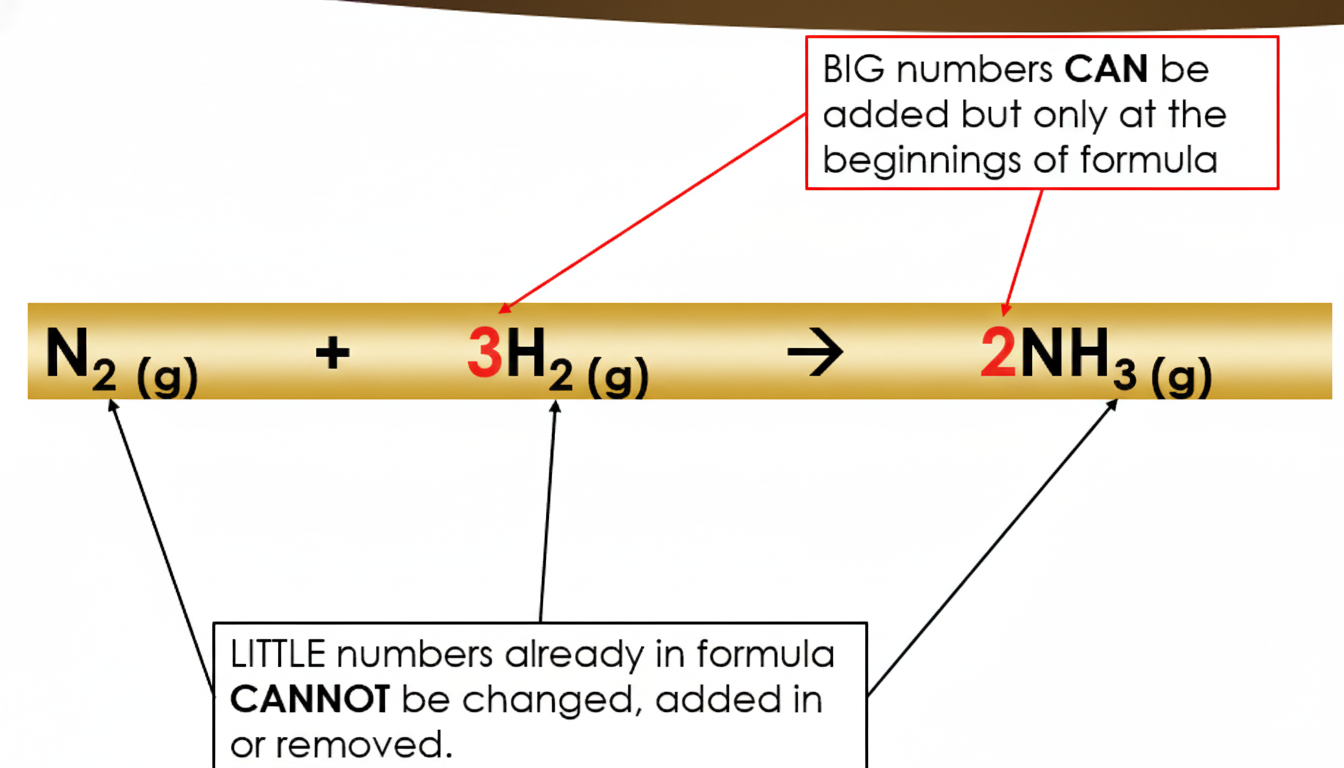
1. You cannot change the element symbols in any formula (for example, you cannot swap an O for an N).
2. You cannot change, add, or remove the small (subscript) numbers inside a formula, because that would turn it into a different substance.
3. The only thing you can do is place a large number (called a coefficient) in front of a whole formula to increase how many of that substance you have.
Reading Chemical Formulae
Before we can successfully learn the skill of balancing, we have to be able to read and break down chemical formulae. We need to be able to count the number of atoms and work out the different types of atoms as shown below:



Counting Atoms Carefully
Once we can count atoms in formulae, we are ready to balance chemical equations. There are a few common traps to watch out for. The symbol for aluminium is Al (one capital letter followed by one lowercase letter). Do not mistake the lowercase "l" for the element iodine (I); the whole symbol Al represents a single element.
Another example is CO compared with Co. CO contains two capital letters, so it represents two separate elements: carbon (C) and oxygen (O). Co has only one capital letter followed by a lowercase letter, so it is a single element: cobalt. The rule is that every element symbol starts with exactly one capital letter, sometimes followed by one lowercase letter.
Balancing Methods
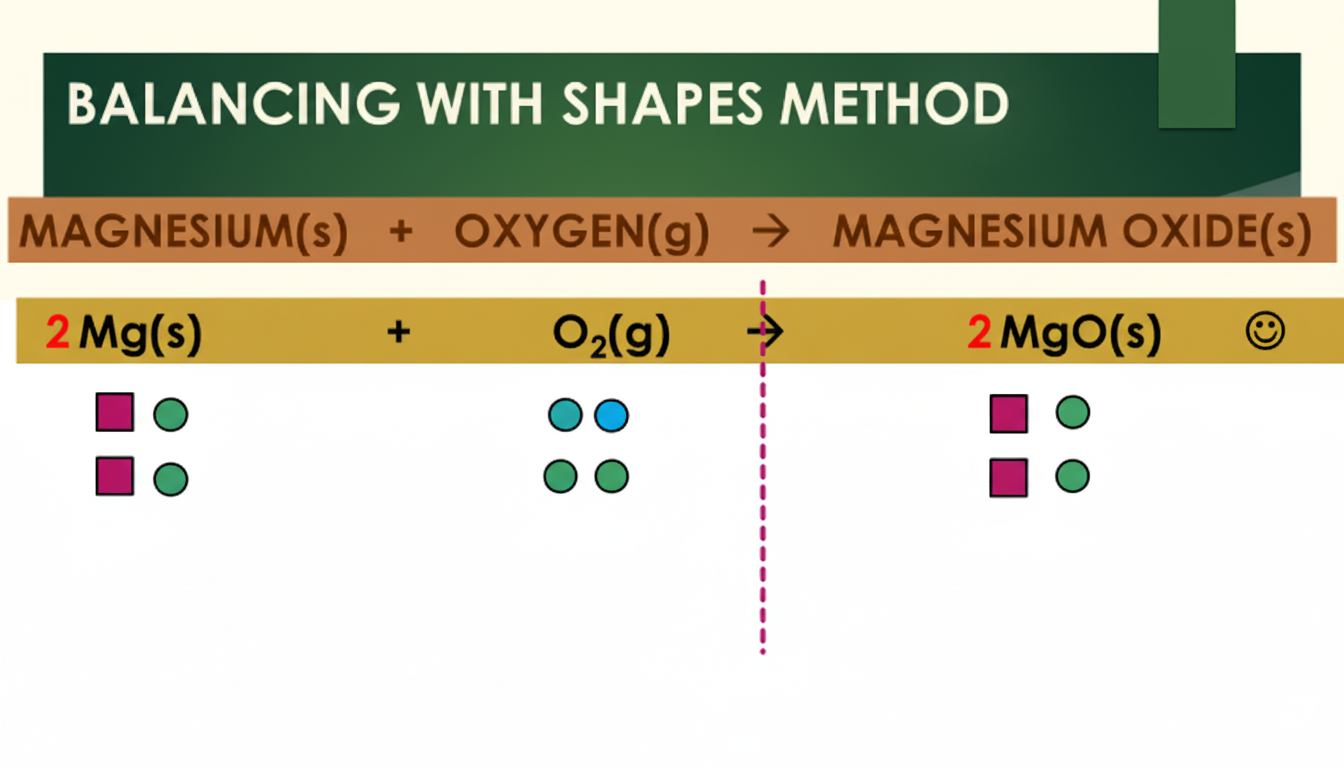
Example 1: Magnesium and Oxygen
This first example shows how magnesium reacts with oxygen to form magnesium oxide. The first image shows how this equation can be balanced with the Symbols Method. The second image shows the same reaction being balanced using the Shapes Method:

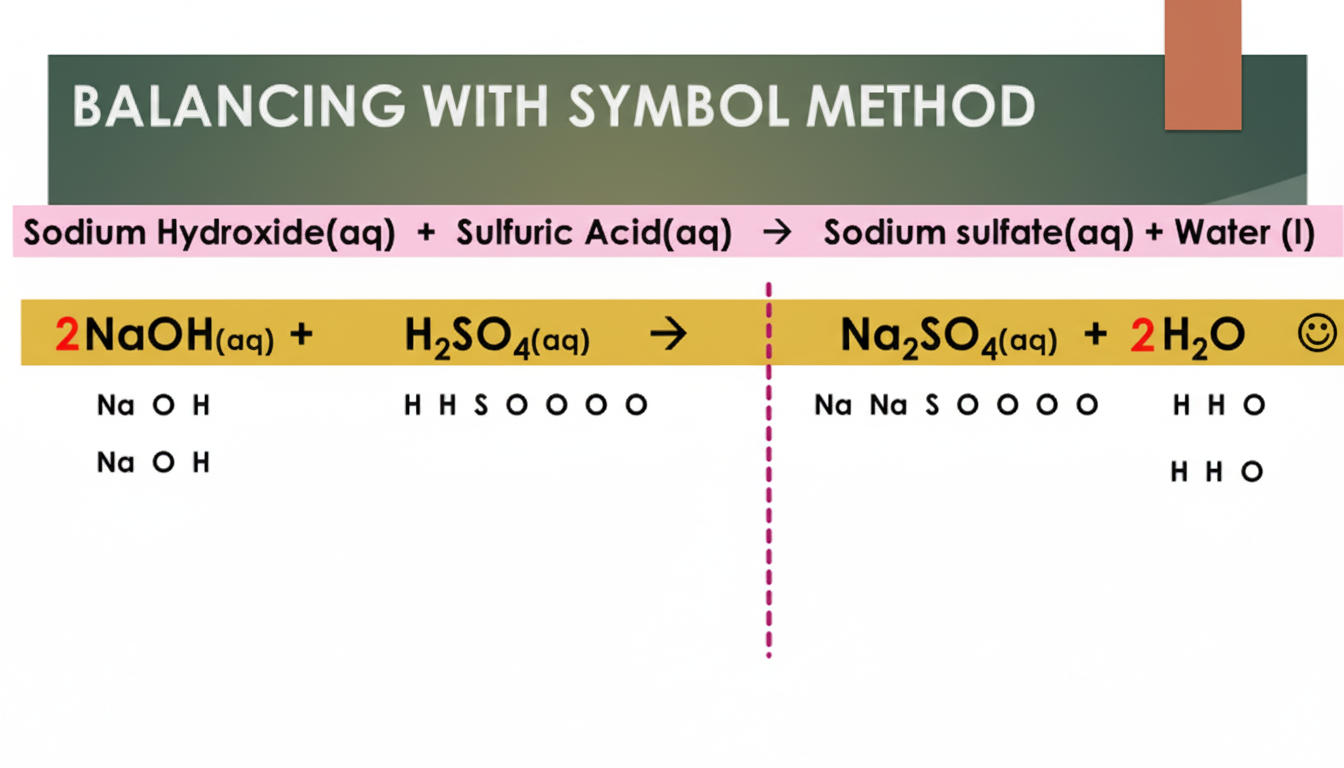
Example 2: Sodium Hydroxide and Sulfuric Acid
This second example shows how sodium hydroxide (NaOH) reacts with sulfuric acid (H2SO4) to form sodium sulfate and water. The first image shows how this equation can be balanced with the Symbols Method. The second image shows the same reaction being balanced using the Shapes Method:
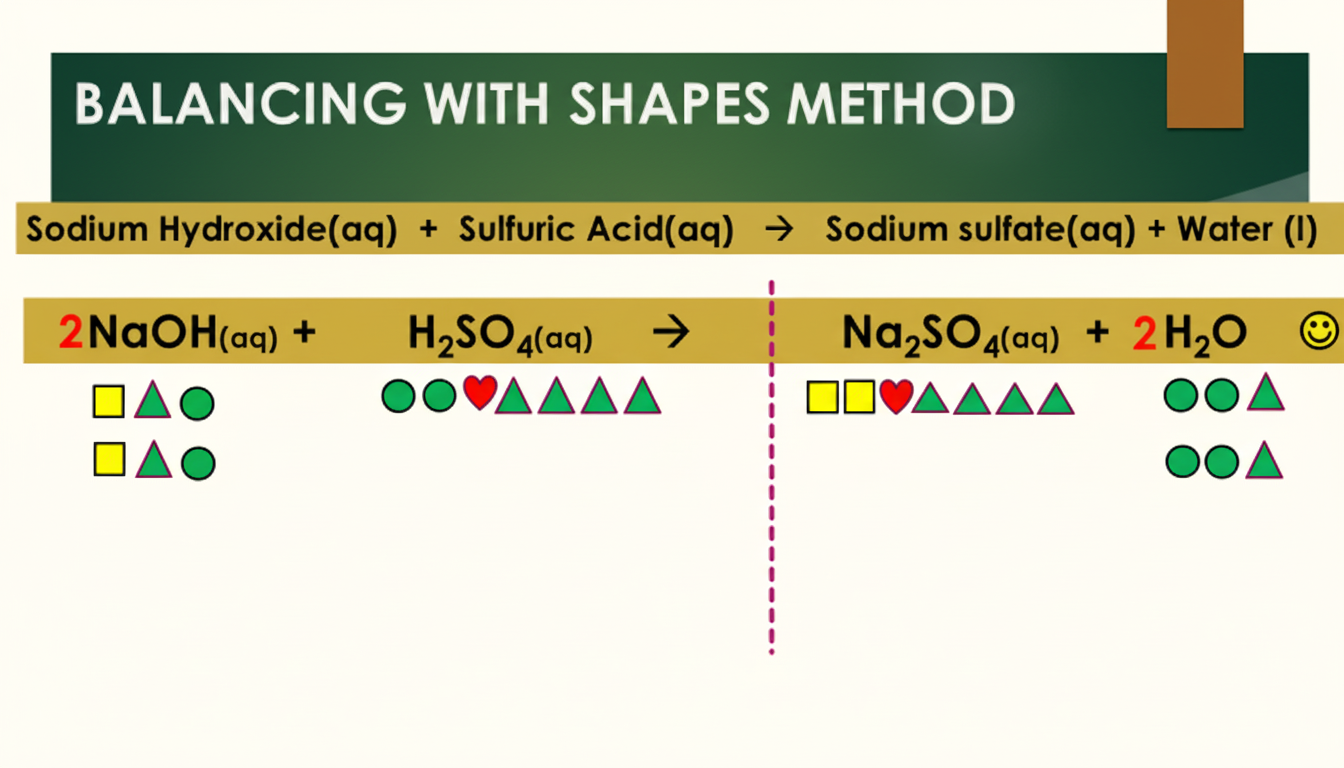
Whichever method you use, the principle is the same: count the atoms of each element on both sides. If they do not match, add big numbers in front of the formulae until they do. Never change the small (subscript) numbers, as that would change the substance itself.
Summary
- Chemical equations must be balanced so that the number of atoms of each element is the same on both sides. This follows the Law of Conservation of Mass.
- Only big coefficients in front of formulae can be changed. Symbols and subscript numbers must stay as they are.
- Before balancing, practise reading formulae and counting the total number of each type of atom.
- Watch out for elements with two-letter symbols (e.g. Al, Co, Na) and do not confuse them with two separate elements.
- You can balance using the Symbols Method or the Shapes Method. Try both and use whichever you find easiest.
Related Lessons
- Atomic Structure – understand how to read the periodic table and work out the number of subatomic particles in an atom.
- Moles & Avogadro’s Constant – learn how to use balanced equations alongside mole calculations.