Video Lesson
Introduction
This lesson covers the fundamentals of atomic structure, which are required by all GCSE Chemistry examination boards. We will look at how to use the periodic table to work out how many subatomic particles an atom of a particular element has, and how to draw atoms.
Using the Periodic Table
For GCSE Chemistry, we need to use the periodic table to extract information about atoms. The periodic table shown below is a good example of the type used at GCSE:
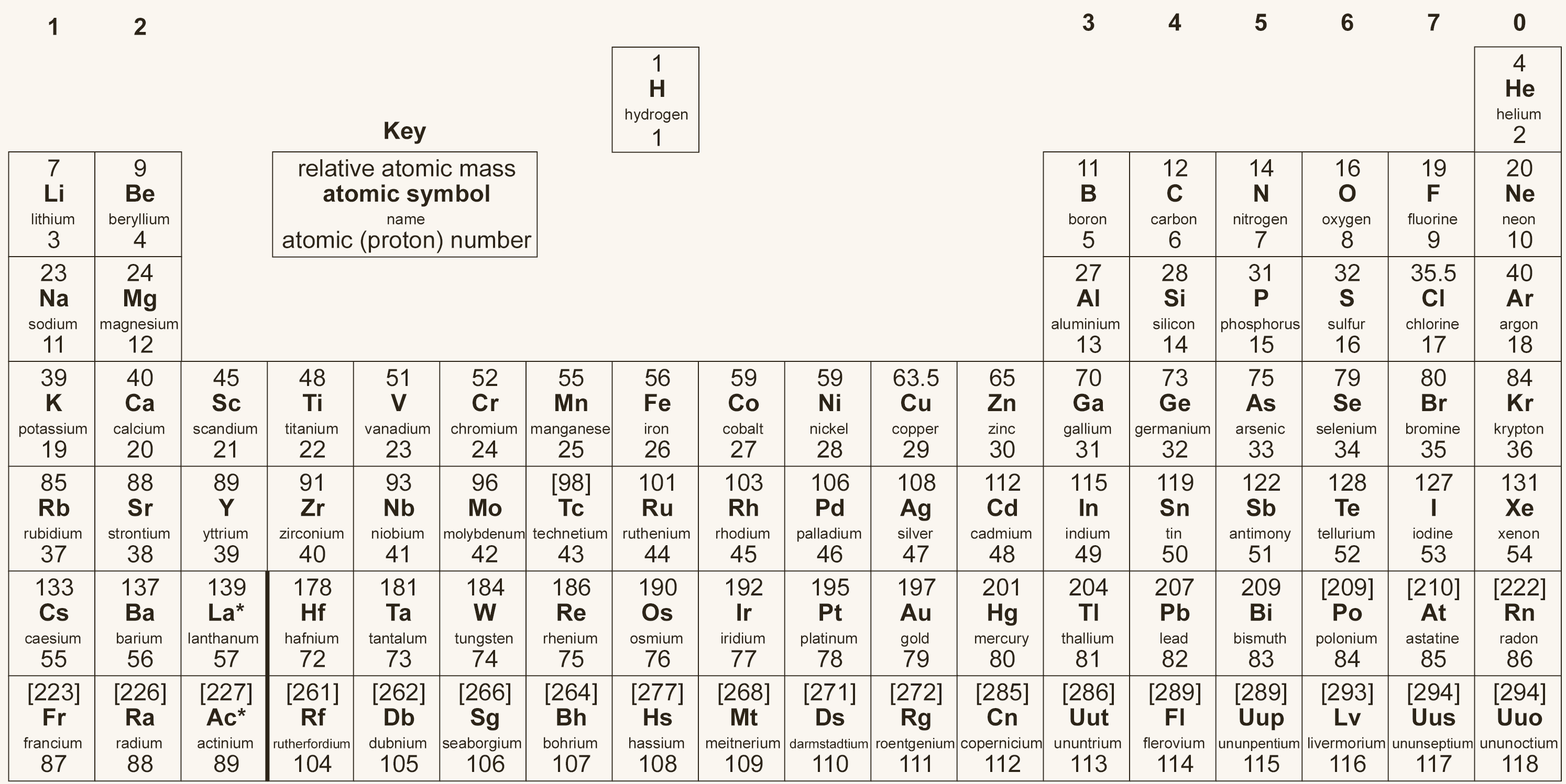
Each element symbol has two numbers associated with it. The most important is the atomic number (the smaller number), because this defines the element. For example, only beryllium has 4 protons, and if an unknown element has 27 as its atomic number, that element must be cobalt.
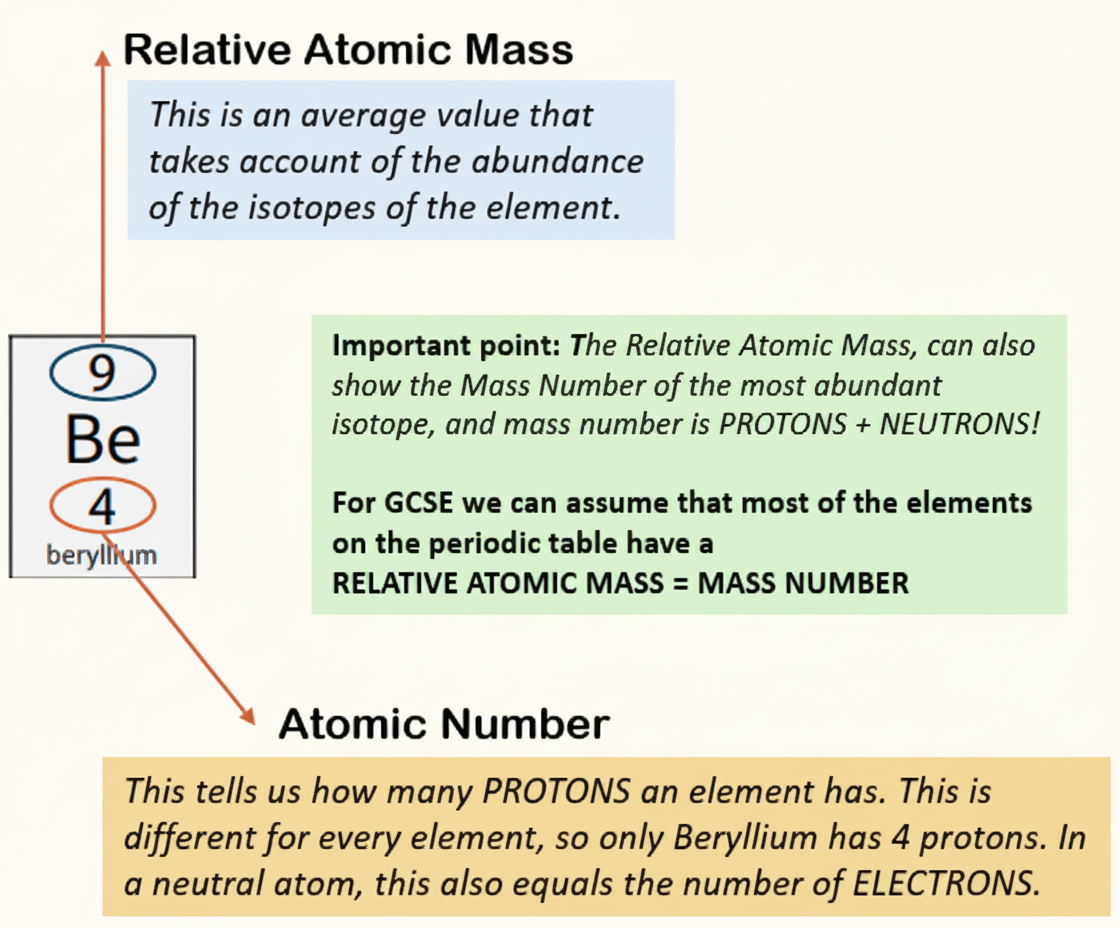
The other number is the relative atomic mass, which is the average mass of all the isotopes of that element. Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
Isotopes: Bromine Example
An example of this is bromine. Bromine has two isotopes of equal abundance, one with a mass number of 79 and the other with a mass number of 81:
| Isotope | Number of Protons | Number of Neutrons | Abundance (%) |
|---|---|---|---|
| Bromine-79 | 35 | 44 | 50 |
| Bromine-81 | 35 | 46 | 50 |
The average value for these two isotopes is 80, so bromine has a relative atomic mass of 80.
Isotopes: Carbon Example
For most elements, the isotopes are not of equal abundance, so there is usually one isotope that is the most abundant. A great example of this is carbon, which has three isotopes:
| Isotope | Number of Protons | Number of Neutrons | Abundance (%) |
|---|---|---|---|
| Carbon-12 | 6 | 6 | 98.9 |
| Carbon-13 | 6 | 7 | 1.1 |
| Carbon-14 | 6 | 8 | <0.1 |
Because the most abundant isotope for carbon is Carbon-12, the GCSE periodic table lists 12 as the relative atomic mass. For GCSE, we can assume that the relative atomic mass of an element is also the mass number of the most common isotope.
Working Out Subatomic Particles
We can use the information from the periodic table to work out the number of subatomic particles in an atom. Let us look at carbon:
| Subatomic Particle | Carbon |
|---|---|
| Number of Protons | 6 |
| Number of Electrons | 6 |
| Number of Neutrons | 6 |
The number 12 represents the relative atomic mass (in this case also the mass number) of an atom of carbon. The mass number tells us how many protons and neutrons are found in the nucleus.
The number 6 represents the atomic number of an atom of carbon. The atomic number tells us carbon has 6 protons. It also tells us carbon has 6 electrons, because in a neutral atom the number of protons must equal the number of electrons.
Finally, as we know the number of protons is 6, and we know that the mass number is 12, we can subtract the atomic number from the mass number to work out the number of neutrons: 12 − 6 = 6 neutrons.
Protons = atomic number.
Electrons = atomic number (in a neutral atom).
Neutrons = mass number − atomic number.
More Examples
Here are a few more examples. These are the answers to the examples in the video lesson:
Sodium (Na)
| Subatomic Particle | Sodium |
|---|---|
| Number of Protons | 11 |
| Number of Electrons | 11 |
| Number of Neutrons | 12 (23 − 11 = 12) |
Aluminium (Al)
| Subatomic Particle | Aluminium |
|---|---|
| Number of Protons | 13 |
| Number of Electrons | 13 |
| Number of Neutrons | 14 (27 − 13 = 14) |
Titanium (Ti)
| Subatomic Particle | Titanium |
|---|---|
| Number of Protons | 22 |
| Number of Electrons | 22 |
| Number of Neutrons | 26 (48 − 22 = 26) |
Electronic Structure
The periodic table symbols can also be used to draw atoms. It is important to remember that protons and neutrons are found in the nucleus and electrons are found in the shells. The shells are also sometimes called energy levels. Shells can only be filled in a particular order. You must start by filling the shell closest to the nucleus first. The first shell can hold a maximum of 2 electrons, the second shell can hold a maximum of 8, and the third shell can also hold a maximum of 8 electrons (at GCSE level).
We can also express the electron arrangement as a number code. If, for example, we have 13 electrons, the electrons fill as follows: 2 in the first shell, 8 in the second shell, and the remaining 3 go into a third shell. This can be written as 2,8,3. This is called the electronic structure (or electronic configuration).
| Element Name | Number of Electrons | Electronic Structure | Group Number |
|---|---|---|---|
| Boron | 5 | 2,3 | 3 |
| Carbon | 6 | 2,4 | 4 |
| Silicon | 14 | 2,8,4 | 4 |
| Potassium | 19 | 2,8,8,1 | 1 |
| Sulfur | 16 | 2,8,6 | 6 |
Linking Electronic Structure to the Periodic Table
It is also important to realise that when you have the electronic structure code, the last number represents the number of electrons in the outer shell. This also corresponds to the group number. A group is a column on the periodic table. We can therefore say that:
- Number of electrons in outer shell = Group number
- Number of shells = Period number
Example
Aluminium is in Group 3 and Period 3. Aluminium has an atomic number of 13, so it has 3 electron shells: 2 in the first shell, 8 in the second shell, and 3 in the outer shell. The number of outer-shell electrons matches its group number of 3.
Drawing Atoms
We can use the information found on the element's symbol to help us draw atom diagrams. We can also use the position of the symbol in the periodic table to check we have done everything correctly:
| Subatomic Particle | Aluminium |
|---|---|
| Number of Protons | 13 |
| Number of Electrons | 13 |
| Number of Neutrons | 14 (27 − 13 = 14) |
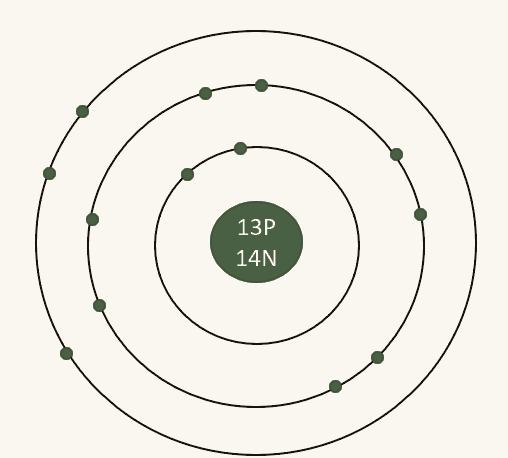
Here is a diagram of aluminium. We know that protons and neutrons are found in the nucleus, and we have worked out how many of each subatomic particle there are from the element's symbol. Aluminium has 13 electrons, drawn across 3 shells as shown. We can count a total of 13 electrons over 3 shells (matching Period 3), with 3 electrons in the outer shell (matching Group 3). That confirms the diagram is correct.
Always use the periodic table position to double-check your atom diagram. The period number tells you how many shells to draw, and the group number tells you how many electrons should be in the outer shell.
Summary
- The atomic number (smaller number) tells you how many protons an atom has. In a neutral atom, this also equals the number of electrons.
- The relative atomic mass (larger number) can be used as the mass number for the most abundant isotope at GCSE level.
- Neutrons = mass number − atomic number.
- Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
- Electrons fill shells in order: 2, then 8, then 8 (at GCSE level).
- The electronic structure links directly to the periodic table: the last number matches the group number, and the number of shells matches the period number.
Related Lessons
- Ions & Chemical Formulae – learn how atoms form ions and how to work out chemical formulae from ion charges.
- Moles & Avogadro’s Constant – use relative atomic mass to convert between moles, mass, and number of particles.